Research in Organic Chemistry

Minehan Group
Back From left: Ashley
Salvador, Daljeet Kaur, Michael Khoury, Mikayla Patterson, Donald Eli Trujillo,
Aren Mirzakhanian, Jose Salvatierra, Fred Farrar, Karen Melendez
Edward Brian Huseman, Kapil Luthra; Front From
Left Natalie Silberberg, Vanessa Medina, Philip Chung, James Nguyen
Research
in the Minehan group is primarily focused in three areas: 1. The total
synthesis of biologically active natural products; 2. The development of new
synthetic methods for carbon-carbon bond-formation, with an emphasis on
advancing the utility of ynol ethers or ynolates as synthetic intermediates; 3.
The development of sequence-specific major–groove binding small molecules for
the regulation of gene expression. We are currently pursuing four main lines of
investigation:
1.
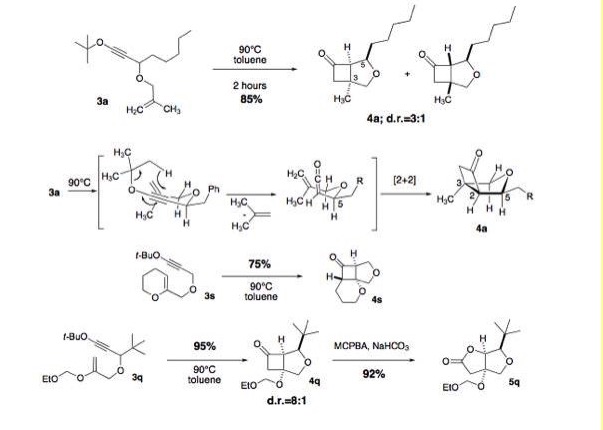
The [3,3]-sigmatropic rearrangement of aliphatic
allyl-alkynyl ethers is a relatively unexplored reaction in organic synthesis.
Allyl-alkynyl ethers may be generated from allyl-1,1-dichlorovinyl ethers by
treatment with excess n-BuLi at low temperatures; however, subjecting
allyl-dichlorovinyl ethers to these conditions, followed by quenching with an
alcohol, leads to rearranged products in the form of gamma, delta-unsaturated
esters. Sigmatropic rearrangement is occurring rapidly and stereospecifically
at low temperatures in this process. Mechanistic investigations of this
reaction are underway; the proposed ketene intermediate holds great synthetic
promise, since it may be intercepted by a wide variety of nucleophiles to form
an array of carbonyl compounds in a single step. We have recently developed
milder methods for the preparation of alkynyl ethers and have successfully
applied these methods in the rearrangement process.

2.
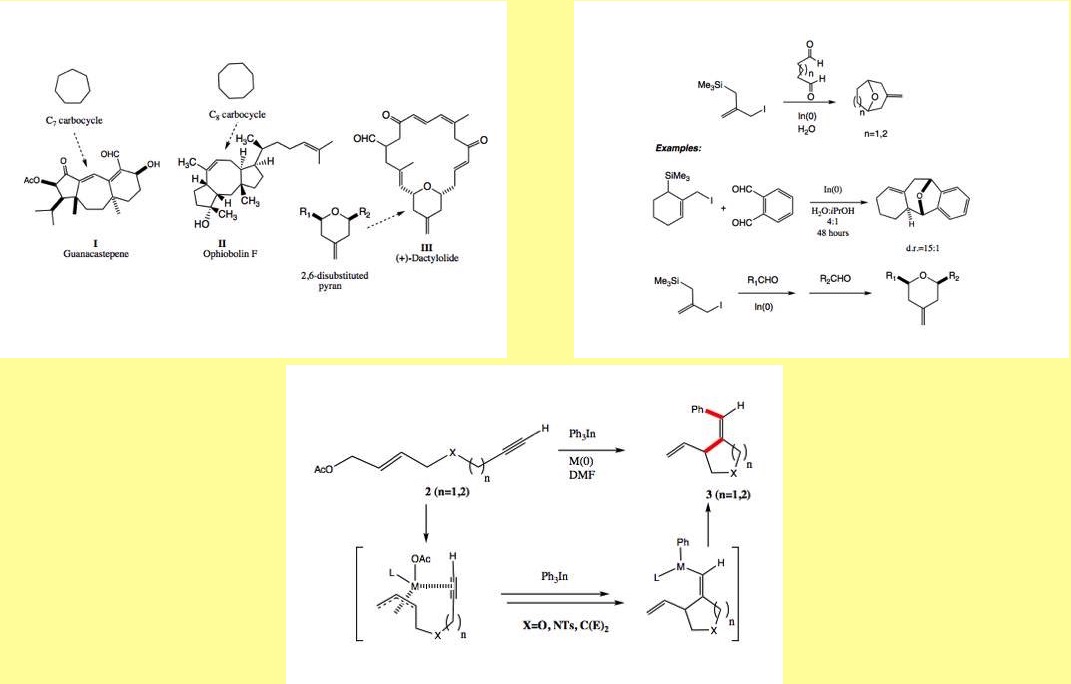
Allylindium reagents, generated in-situ from allyl
halides and indium metal, react with carbonyl compounds in water to form
products containing a new carbon-carbon bond. We have prepared a series of
silyl-substituted allylindium reagents that allow multiple carbon-carbon bonds
to be formed in a single step in aqueous media upon reaction with appropriate
electrophiles. Such reagents may be employed in an environmentally benign
preparation of substituted pyrans and 7- and 8-membered carbocycles, which are at
the core of a variety of biologically-important natural products. Allylic
substitution reactions are important and powerful methods for carbon-carbon
bond-formation. Environmentally benign organoindium reagents participate in
transition-metal catalyzed cross-coupling reactions and allylic substitution
reactions.
We have
reported that 1-acetoxy-2,7- and 2,8-enynes undergo a palladium-catalyzed
cyclization / substitution reaction in the presence of organoindium reagents to
form substituted 5- and 6-membered carbo- and heterocycles. We envision that
this process may be extended to the stereoselective synthesis of fused 6-5 ring
systems found in many natural products.
3.
C-aryl
glycosides are an important class of naturally occurring compounds with unique
chemical and biological properties.
Possessing a carbon-carbon bond between aromatic and carbohydrate moieties,
these substances are endowed with remarkable stability toward acid and
enzymatic hydrolysis; this affords them sufficient intracellular lifetime to
allow trafficking to the nucleus, where they bind DNA to form stable complexes.
Indeed, numerous members of the glycosyl arene family have been show to possess
antibacterial, antitumor, and antifungal activities. We have embarked on a
program directed toward the total synthesis of members of this family of
natural products, and have developed new methods for their synthesis. In
particular, we have recently reported an indium allylation/organoindium cross
coupling route to 2-deoxy C-aryl
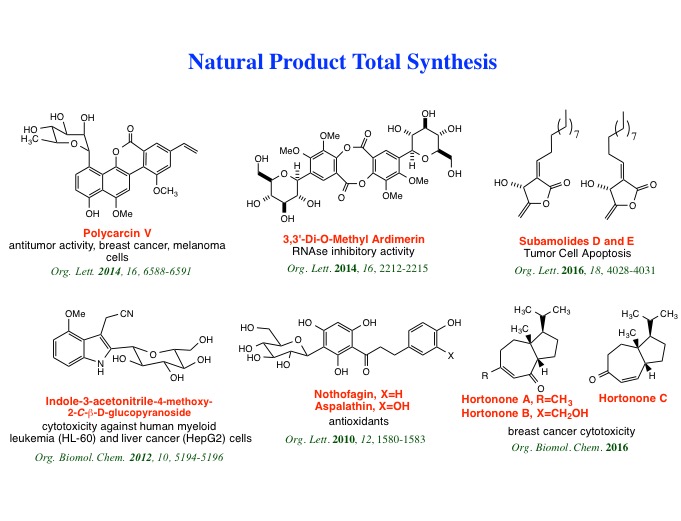
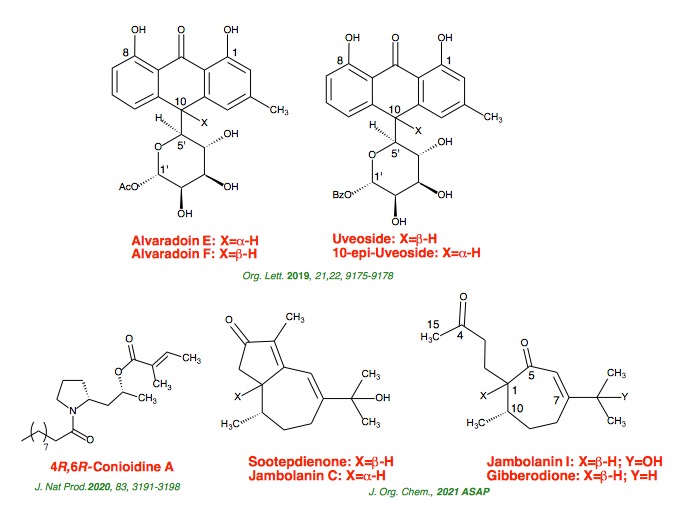
ribofuranosides and pyranosides. Furthermore, we have accomplished the total
syntheses of aspalathin, nothofagin, indole-3-acetonitrile-4-methoxy-2-C-beta-D-glucopyranoside, polycarcin V, 3,3’-di-O-methylardimerin, hortonones A-C,
subamolides D and E, alvaradoins E and F, uveoside and 10-epi-uveoside,
conioidine A, sootepdienone, jambolanins C and I, and gibberodione.
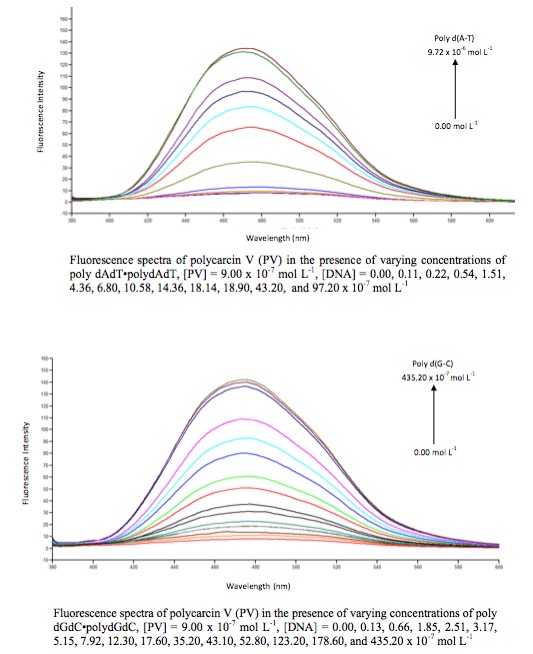
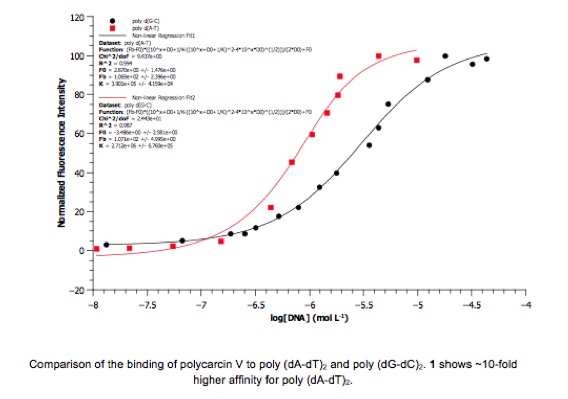
4. We are also assessing the binding affinity
and sequence selectivity of C-aryl
glycosides for duplex nucleic acids using fluorescence and ultraviolet
spectroscopies. Polycarcin V, which has an association constant for calf-thymus
DNA of 1.7(±0.1) x 106M-1, has been found to bind AT-rich
DNA with an order of magnitude greater binding affinity than GC-rich DNA. In
order to further explore the principles of carbohydrate-DNA molecular
recognition, we are currently investigating the preparation bis-C-glycoside derivatives of
intercalating chromophores that place carbohydrate moieties in both the major
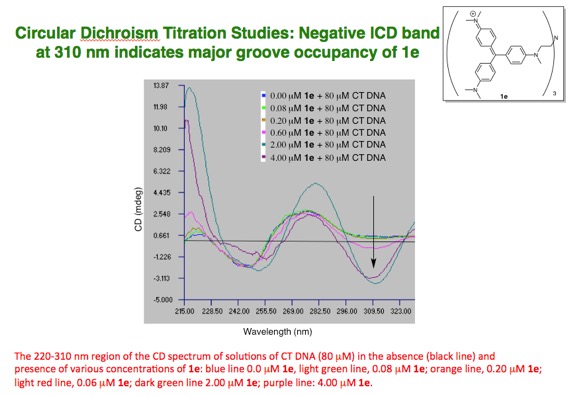
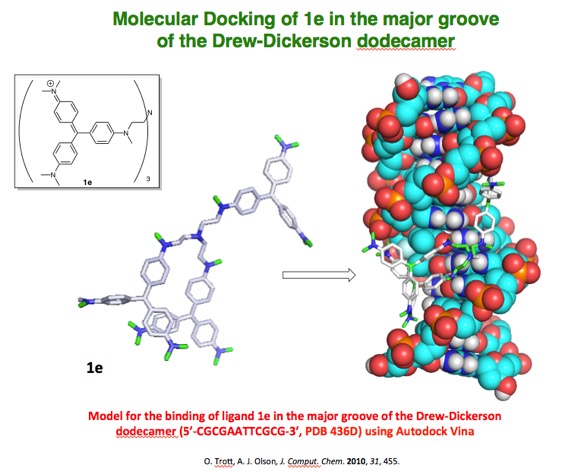
and minor grooves of DNA. Recently we have discovered that derivatives of the
triarylmethane dye crystal violet are non-intercalating major-groove binding
molecules which display a preference for associating with non-alternating AT
tracts of B* form DNA due their steric bulk. Further studies looking
at shape-selective binding of these molecules are currently underway.
Students
in the Minehan group gain practical experience in the synthesis, purification,
and spectroscopic characterization of organic molecules. Some examples of the
useful skills obtained are:
1.
Techniques for running reactions under inert atmosphere
2.
Purification of compounds by
column chromatography and radial chromatography
3.
Analysis of reactions by thin-layer chromatography and GC-MS
4.
Structure elucidation of synthetic compounds using one- and
two-dimensional NMR spectroscopy
5.
Characterization of synthetic compounds by LC-MS, IR, and UV
spectroscopies
6.
Evaluation and quantitation of small molecule-macromolecule binding
interactions by UV, CD, and fluorescence spectroscopy
Useful
website for beginning laboratory students: Synthetic organic laboratory
techniques
NSF-Funded
Research Descriptive Video
Research
Funding
National Science Foundation
National Institutes of Health
American Chemical Society Petroleum
Research Fund
Henry Dreyfus Teacher Scholar Award
Research Corporation
CSUN Competition for Research, Scholarship
and Creative Activity Award
Organic Syntheses, Wayland E Noland
Undergraduate Research Grant
CSUPERB Faculty-Student Collaborative
Grant
Selected Recent Publications:
21. Total
Synthesis of Indole-3-Acetonitrile-4-Methoxy-2-C-b-D-Glucopyranoside.
Proposal for Structural Revision of the Natural Product Yepremyan,
A.; Minehan, T.G. Org. Biomol. Chem. 2012, 10, 5194-5196.
23. [3,3]-Sigmatropic
Rearrangement/5-Exo-Dig Cyclization Reactions of Benzyl Alkynyl Ethers:
Synthesis of Substituted 2-Indanones and Indenes Tudjarian,
A.A.; Minehan, T.G. J. Org. Chem. 2011, 76, 3576-3581.
24. Indium-Mediated Allylation of Aldehydes, Ketones, and
Sulfonimines with 2-(Alkoxy)allyl bromides Dhanjee, H.;
Minehan, T.G. Tetrahedron Lett. 2010, 51, 5609-5612.

Armen
Nazarian and Sayuri Pacheco at the ACS National Meeting in San Francisco, April
2017

Sayuri
Pacheco at the Sigma Xi International Research Conference, Atlanta, Georgia,
November, 2016
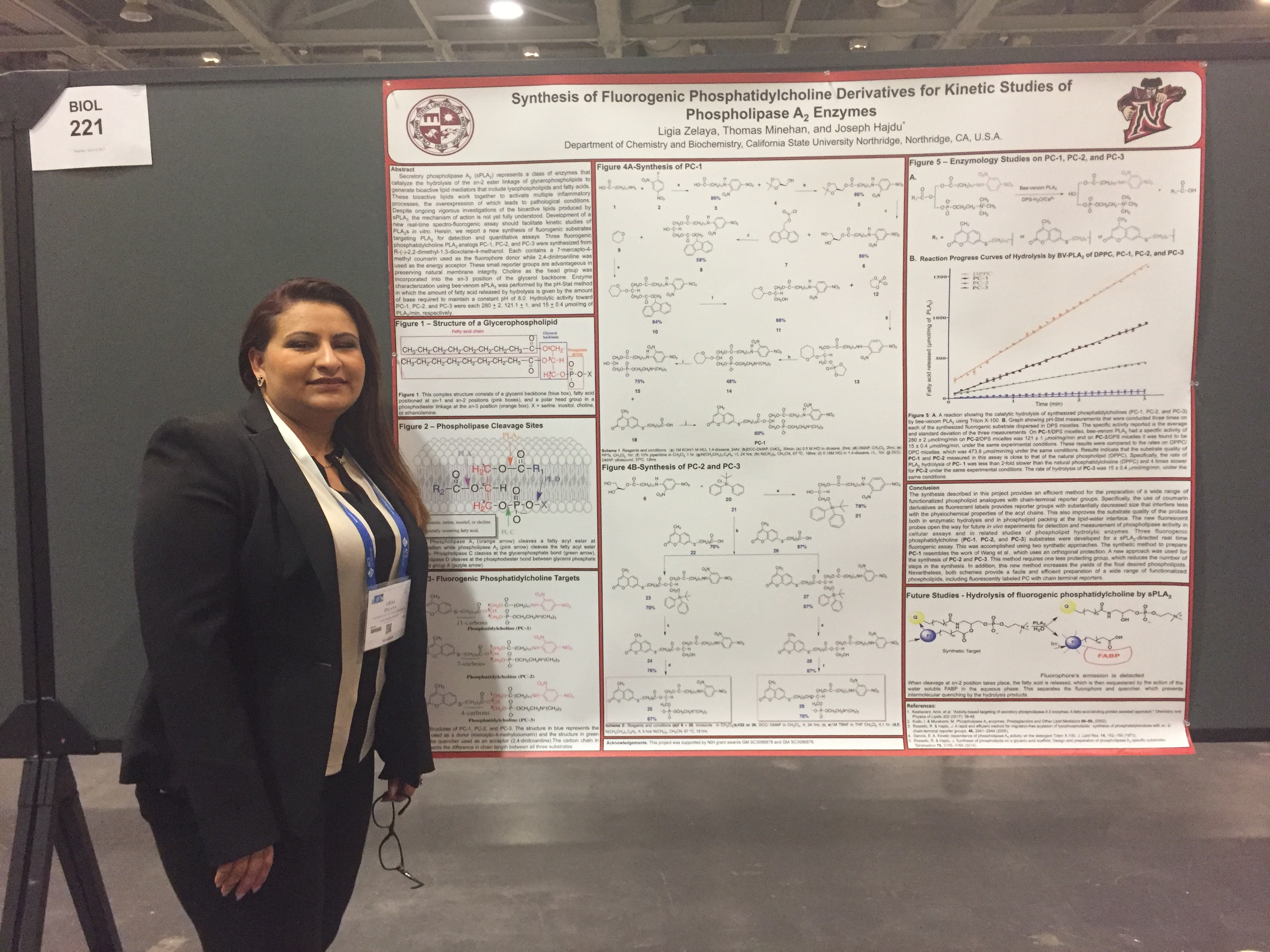
Ligia Zelaya at the 2017
ACS National Meeting in San Francisco

Alejandra Fausto and Bianca Chavez at the 2017
CSUNposium

Kevin Ng at the
2018 ACS National Meeting in Boston, August 2018

Omar Nunez at
the 2018 ACS National Meeting in Boston, August 2018